Group B Streptococcus Nucleic Acid
Product name
HWTS-UR027-Group B Streptococcus Nucleic Acid Detection Kit(Fluorescence PCR)
HWTS-UR028-Freeze-dried Group B Streptococcus Nucleic Acid Detection Kit(Fluorescence PCR)
Certificate
CE, FDA
Epidemiology
Group B Streptococcus (GBS), also known as streptococcus agalactiae, is a gram-positive opportunistic pathogen that normally inhabits the lower gastrointestinal and urogenital tracts of the human body. Approximately 10%-30 % of pregnant women have GBS vaginal sojourn.
Pregnant women are susceptible to GBS infection due to changes in the internal environment of the reproductive tract due to changes in hormone levels in the body, which will cause adverse pregnancy outcomes such as preterm labor, premature rupture of membranes, and stillbirth, and can also lead to puerperal infections in pregnant women.
Neonatal group B streptococcus is associated with perinatal infection and is an important pathogen of severe infectious diseases such as neonatal sepsis and meningitis. 40%-70% of mothers infected with GBS will transmit GBS to their newborns during delivery through the birth canal, causing serious neonatal infectious diseases such as neonatal sepsis and meningitis. If the newborns carry GBS, about 1%-3% will develop early invasive infection, of which 5% will result in death.
Channel
| FAM | GBS target |
| VIC/HEX | Internal control |
Technical Parameters
| Storage | Liquid: ≤-18℃ in dark; Lyophilization: ≤30℃ in dark |
| Shelf-life | 12 months |
| Specimen Type | Genital and Rectal Secretions |
| Ct | ≤38 |
| CV | ≤5.0% |
| LoD | 1×103Copies/mL |
| Covering Subtypes | Detect group B streptococcus serotypes (I a, I b, I c, II, III, IV, V, VI, VII, VIII, IX and ND) and the results are all positive. |
| Specificity | Detect other genital tract and rectal swab samples such as candida albicans, trichomonas vaginalis, chlamydia trachomatis, ureaplasma urealyticum, neisseria gonorrhoeae, mycoplasma hominis, mycoplasma genitalium, herpes simplex virus, human papilloma virus , lactobacillus, gardnerella vaginalis, staphylococcus aureus, national negative reference N1-N10 (streptococcus pneumoniae, streptococcus pyogenes, streptococcus thermophilus, streptococcus mutans, streptococcus pyogenes, lactobacillus acidophilus bacillus, lactobacillus reuteri, escherichia coli DH5α, candida albicans) and human genomic DNA, the results are all negative for group B streptococcus. |
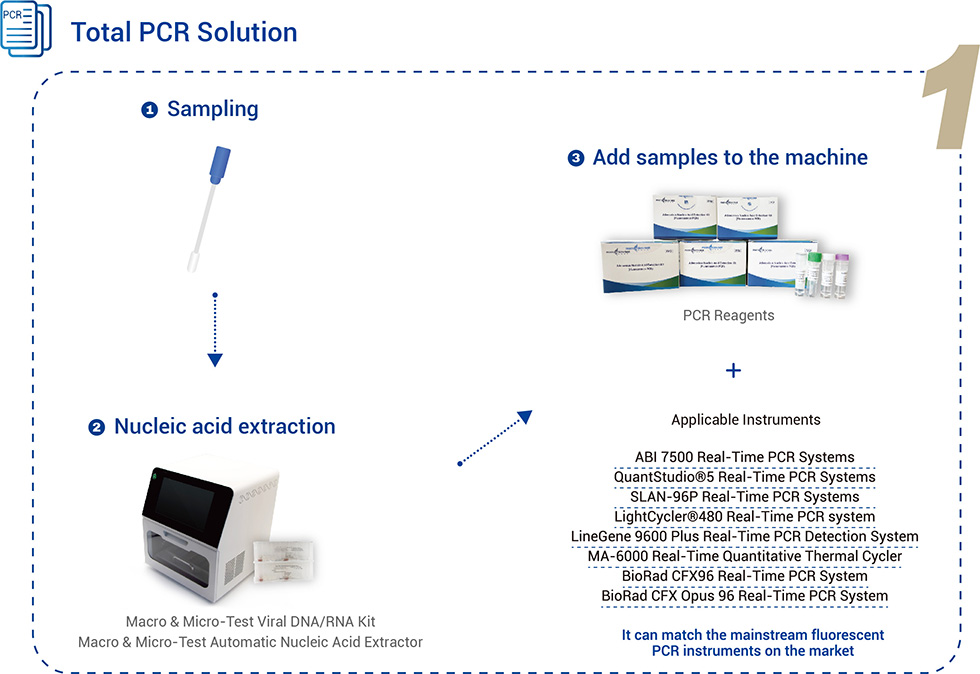
| Applicable Instruments | It can match the mainstream fluorescent PCR instruments on the market. SLAN-96P Real-Time PCR Systems ABI 7500 Real-Time PCR Systems QuantStudio®5 Real-Time PCR Systems LightCycler®480 Real-Time PCR Systems LineGene 9600 Plus Real-Time PCR Detection Systems MA-6000 Real-Time Quantitative Thermal Cycler |
Total PCR Solution