Monkeypox Virus and Typing Nucleic Acid
Product name
HWTS-OT202-Monkeypox Virus and Typing Nucleic Acid Detection Kit (Fluorescence PCR)
Certificate
CE
Epidemiology
Monkeypox (Mpox) is an acute zoonotic infectious disease caused by Monkeypox Virus (MPXV). MPXV is round-bricked or oval in shape, and is a double-stranded DNA virus with a length of about 197Kb[1]. The disease is mainly transmitted by animals, and humans could become infected by being bitten by infected animals or by direct contact with the blood, body fluids and rash of infected animals. The virus can also be transmitted between people, primarily through respiratory droplets during prolonged, direct face-to-face contact or through direct contact with a patient's body fluids or contaminated objects[2-3]. Studies have shown that MPXV forms two distinct clades: clade I (previously known as the Central African clade or Congo Basin clade) and clade II (previously called the West African clade). The mpox of Congo Basin clade has been clearly shown to be transmissible between humans and can cause death, while the mpox of West African clade causes milder symptoms and has a lower rate of human-to-human transmission[4].
The test results of this kit are not intended to be the sole indicator for the diagnosis of MPXV infection in patients, which must be combined with the patient's clinical characteristics and other laboratory test data to correctly judge the infection of the pathogen and formulate a reasonable treatment plan to ensure safe and effective treatment.
Channel
| FAM | MPXV clade II |
| ROX | MPXV universal nucleic acid |
| VIC/HEX | MPXV clade I |
| CY5 | internal control |
Technical Parameters
| Storage |
≤-18℃ |
| Shelf-life | 12 months |
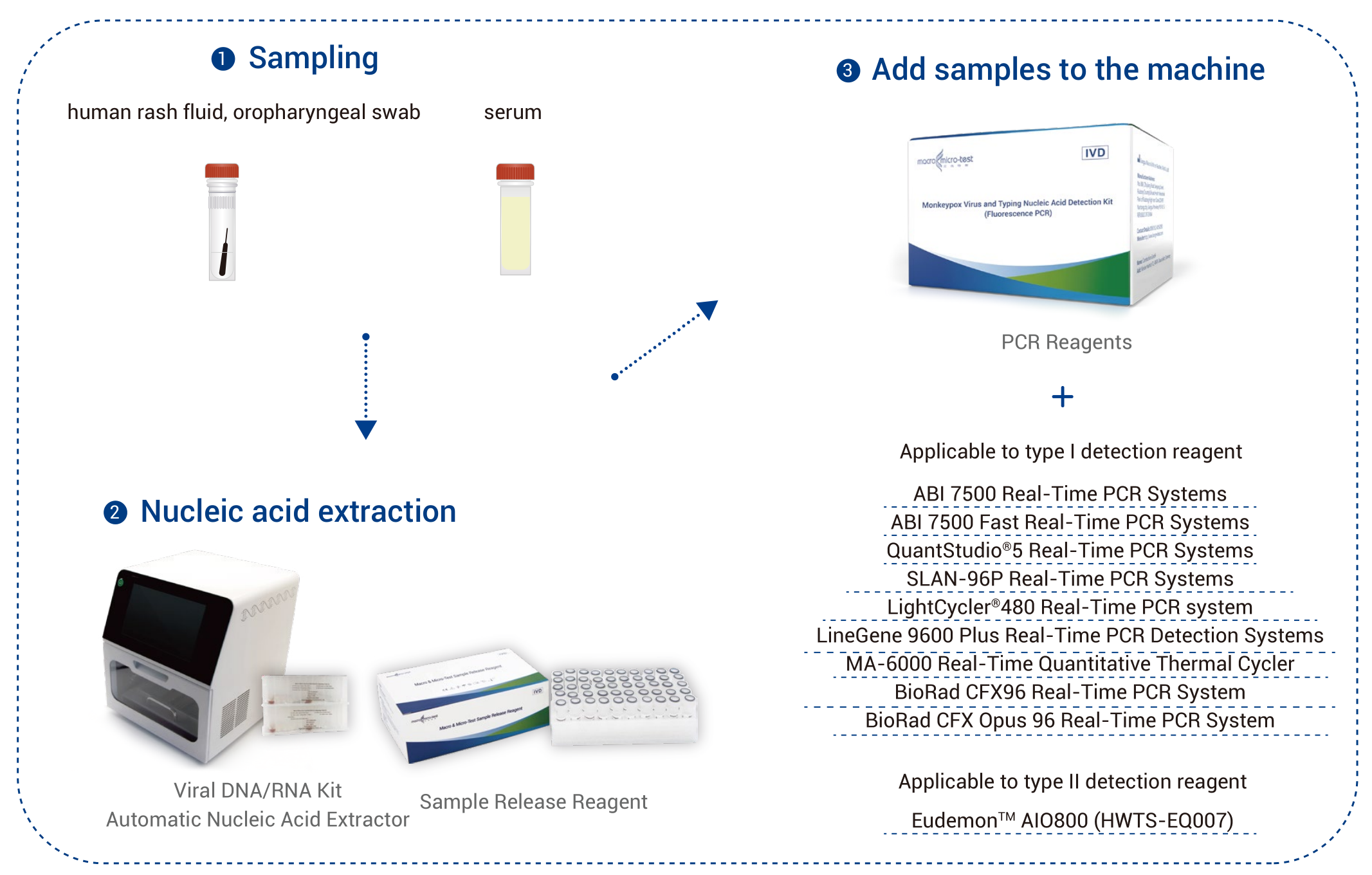
| Specimen Type | human rash fluid, oropharyngeal swabs and serum |
| Ct | ≤38 (FAM, VIC/HEX, ROX), ≤35(IC) |
| LoD | 200 Copies/mL |
| Applicable Instruments | Type I detection reagent:
Applied Biosystems 7500 Real-Time PCR Systems Applied Biosystems 7500 Fast Real-Time PCR Systems QuantStudio®5 Real-Time PCR Systems SLAN-96P Real-Time PCR Systems (Hongshi Medical Technology Co., Ltd.) LightCycler®480 Real-Time PCR system LineGene 9600 Plus Real-Time PCR Detection Systems (FQD-96A, Hangzhou Bioer technology) MA-6000 Real-Time Quantitative Thermal Cycler (Suzhou Molarray Co., Ltd.) BioRad CFX96 Real-Time PCR System BioRad CFX Opus 96 Real-Time PCR System Type II detection reagent: EudemonTM AIO800 (HWTS-EQ007)) by Jiangsu Macro & Micro-Test Med-Tech Co., Ltd. |
Work Flow