Mycobacterium Tuberculosis Nucleic Acid and Rifampicin Resistance
Product name
HWTS-RT074B-Mycobacterium Tuberculosis Nucleic Acid and Rifampicin Resistance Detection Kit (Melting Curve)
Certificate
CE
Epidemiology
Mycobacterium tuberculosis , shortly as Tubercle bacillus, TB, is the pathogenic bacterium that causes tuberculosis. Currently, the commonly used first-line anti-tuberculosis drugs include isoniazid, rifampicin and hexambutol, etc. The second-line anti-tuberculosis drugs include fluoroquinolones, amikacin and kanamycin, etc. The new developed drugs are linezolid, bedaquiline and delamani, etc. However, due to the incorrect use of anti-tuberculosis drugs and the characteristics of the cell wall structure of mycobacterium tuberculosis, mycobacterium tuberculosis develops drug resistance to anti-tuberculosis drugs, which brings serious challenges to the prevention and treatment of tuberculosis.
Rifampicin has been widely used in the treatment of pulmonary tuberculosis patients since the late 1970s, and has a significant effect. It has been the first choice to shorten the chemotherapy of pulmonary tuberculosis patients. Rifampicin resistance is mainly caused by the mutation of the rpoB gene. Although new anti-tuberculosis drugs are constantly coming out, and the clinical efficacy of pulmonary tuberculosis patients has also continued to improve, there is still a relative lack of anti-tuberculosis drugs, and the phenomenon of irrational drug use in clinical is relatively high. Obviously, the Mycobacterium tuberculosis in patients with pulmonary tuberculosis cannot be completely killed in a timely manner, which eventually leads to different degrees of drug resistance in the patient's body, prolongs the course of the disease, and increases the risk of death of the patient.
Channel
|
Channel |
Channels and Fluorophores |
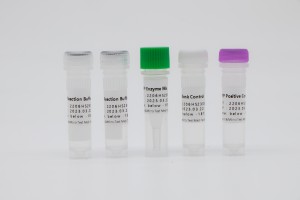
Reaction Buffer A |
Reaction Buffer B |
Reaction Buffer C |
|
FAM Channel |
Reporter: FAM, Quencher: None |
rpoB 507-514 |
rpoB 513-520 |
38KD and IS6110 |
|
CY5 Channel |
Reporter: CY5, Quencher: None |
rpoB 520-527 |
rpoB 527-533 |
/ |
|
HEX (VIC) Channel |
Reporter: HEX (VIC), Quencher: None |
Internal control |
Internal control |
Internal control |
Technical Parameters
|
Storage |
≤-18℃ In dark |
|
Shelf-life |
12 months |
|
Specimen Type |
Sputum |
|
CV |
≤5.0% |
|
LoD |
mycobacterium tuberculosis 50 bacteria/mL rifampicin-resistant wild type: 2x103 bacteria/mL homozygous mutant: 2x103 bacteria/mL |
|
Specificity |
It detects wild-type mycobacterium tuberculosis and the mutation sites of other drug resistance genes such as katG 315G>C\A, InhA-15C> T, the test results show no resistance to rifampicin, which means there is no cross-reactivity. |
|
Applicable Instruments: |
SLAN-96P Real-Time PCR Systems
BioRad CFX96 Real-Time PCR System LightCycler480® Real-Time PCR System |
Work Flow
If use the Macro & Micro-Test General DNA/RNA Kit (HWTS-3019-50, HWTS-3019-32, HWTS-3019-48, HWTS-3019-96) (which can be used with Macro & Micro-Test Automatic Nucleic Acid Extractor (HWTS-3006C, HWTS-3006B)) or Macro & Micro-Test Viral DNA/RNA Column(HWTS-3022-50) by Jiangsu Macro & Micro-Test Med-Tech Co., Ltd. for extraction, add 200μL of the Positive Control, negative control and processed sputum sample to be tested in sequence, and add 10μL of the internal control separately into the Positive Control, negative control and processed sputum sample to be tested, and the subsequent steps should be strictly carried out according to the extraction instructions. The extracted sample volume is 200μL, and the recommended elution volume is 100μL.